An adaptive design clinical trial can help to create a stronger value proposition for your medical device and give you a competitive edge by reducing development costs and accelerating time-to-market. In particular we resize the study and adjust the critical value for hypothesis testing simultaneously after an interim analysis to achieve a target power and control the type I error rate in comparing AUCs of two modalities. Adaptive designs for medical device clinical studies.
Adaptive Designs For Medical Device Clinical Studies, The US Food and Drug Administration FDA on Tuesday finalized guidance that lays out how to design medical device clinical trials that allow for changes based on data while maintaining study validity and integrity. An adaptive design for a medical device clinical study is defined as a clinical study design that allows for prospectively planned modifications based on accumulating study data without. The purpose is to. The pace of the uptake of adaptive designs in clinical research however has remained well behind that of the statistical literature introducing new methods and highlighting their potential advantages.
 Pin On Services From pinterest.com
Pin On Services From pinterest.com
The purpose is to. The most frequently appearing type of adaptation was the seamless Phase IIIII design 81142 57 followed by adaptive group sequential 30142 21 biomarker adaptive 28142 20 adaptive dose-finding 23142 16 pick-the-winnerdrop-the-loser 13142 9 sample size re-estimation 11142 8 adaptive randomisation 10142 7 adaptive. By spelling out when adaptive designs are acceptable in clinical trials for devices requiring Premarket Approval PMA or 510k premarket notification the FDA seeks to better inform both device manufacturers and its own. SMi Groups Adaptive Designs in Clinical Trials Conference will return to London on 3rd and 4th April 2017 and will feature two case studies on Bayesian adaptive designs in medical device development.
The most frequently appearing type of adaptation was the seamless Phase IIIII design 81142 57 followed by adaptive group sequential 30142 21 biomarker adaptive 28142 20 adaptive dose-finding 23142 16 pick-the-winnerdrop-the-loser 13142 9 sample size re-estimation 11142 8 adaptive randomisation 10142 7 adaptive.
Read another article:
Trial planning involves several rounds of simulations. It provides clarity on how to plan and implement adaptive designs for clinical studies used in medical device development. An adaptive design for a medical device clinical study is defined as a clinical study design that allows for prospectively planned modifications based on accumulating study data without. The 33-page guidance which finalizes a draft version released for comment in September 2018 and replaces an earlier guidance from 2010 sets out FDAs recommendations on adaptive trial design principles and the information FDA will review from adaptive studies submitted as part of investigational new drug applications INDs new drug applications. Barnes PJ Pocock SJ Magnussen H et al.
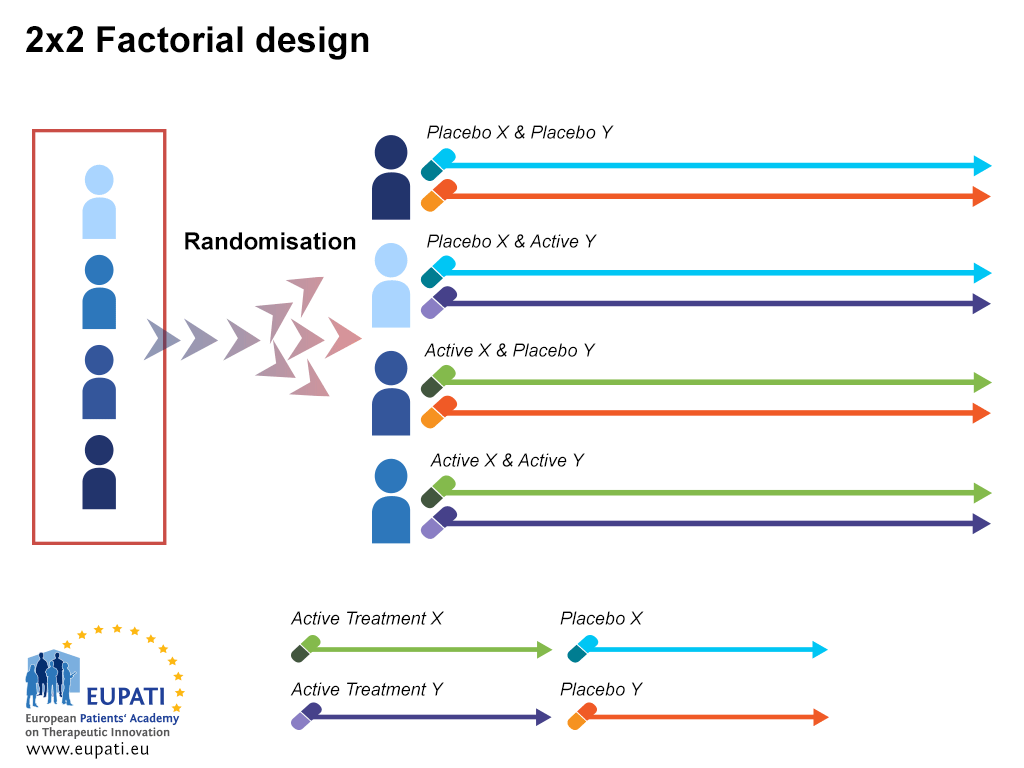 Source: toolbox.eupati.eu
Source: toolbox.eupati.eu
This final guidance applies to premarket medical device submissions including premarket approval applications PMA premarket notification. In essence adaptive designs allow prospectively planned modifications to a clinical trial based on interim data provided scientific validity the ability to draw sound inferences and data integrity credibility and reproducibility are preserved. Bringing in voices from the industrys best clinical specialists strategists and statisticians SMis 9th Adaptive Designs in Clinical Trials conference will feature case. Pulm Pharmacol Ther 2010 23. Clinical Trial Designs Eupati Toolbox.
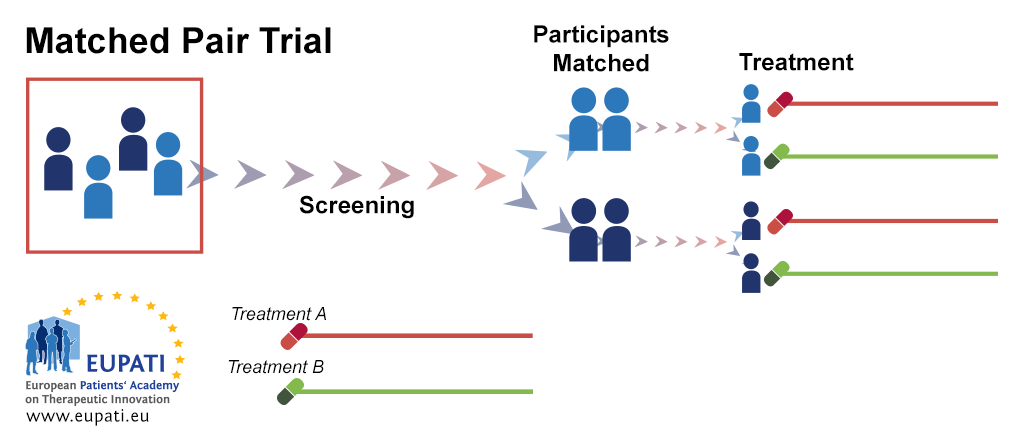 Source: toolbox.eupati.eu
Source: toolbox.eupati.eu
FDA Finalizes Guidance on Adaptive Designs for Device Studies. In essence adaptive designs allow prospectively planned modifications to a clinical trial based on interim data provided scientific validity the ability to draw sound inferences and data integrity credibility and reproducibility are preserved. FDA says it received 151 comments from seven entities including. The pharmaceutical landscape is evolving as stakeholders pursue the use of real-world evidence precision medicine and complex innovative trial designs CID to increase efficiency of development. Clinical Trial Designs Eupati Toolbox.
 Source: clinicaltherapeutics.com
Source: clinicaltherapeutics.com
Bringing in voices from the industrys best clinical specialists strategists and statisticians SMis 9th Adaptive Designs in Clinical Trials conference will feature case. Adaptive designs can be applied across all phases of clinical research from early-phase dose escalation to confirmatory trials. Known as adaptive design the method can minimize clinical trial sponsors resource requirements and increase chances of study success. An adaptive design clinical trial can help to create a stronger value proposition for your medical device and give you a competitive edge by reducing development costs and accelerating time-to-market. The Evolution Of Master Protocol Clinical Trial Designs A Systematic Literature Review Clinical Therapeutics.
 Source: researchgate.net
Source: researchgate.net
The 33-page guidance which finalizes a draft version released for comment in September 2018 and replaces an earlier guidance from 2010 sets out FDAs recommendations on adaptive trial design principles and the information FDA will review from adaptive studies submitted as part of investigational new drug applications INDs new drug applications. Adaptive designs can be applied across all phases of clinical research from early-phase dose escalation to confirmatory trials. There has been considerable interest among pharmaceutical and other medical product developers in adaptive clinical trials in which knowledge learned during the course of a trial affects ongoing conduct or analysis of the trial. Consequences and gains of possible trial adaptations need to be understood before initiation. Summary Of Different Types Of Adaptive Designs For Clinical Trials Download Scientific Diagram.
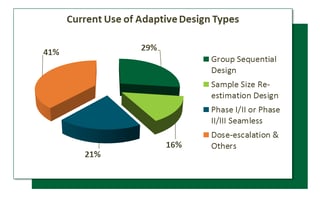 Source: veristat.com
Source: veristat.com
Adaptive designs can be applied across all phases of clinical research from early-phase dose escalation to confirmatory trials. FDA says it received 151 comments from seven entities including. Evaluation of medical imaging devices often involves clinical studies where multiple readers MR read images of multiple cases MC for a clinical task which are often called MRMC studies. In essence adaptive designs allow prospectively planned modifications to a clinical trial based on interim data provided scientific validity the ability to draw sound inferences and data integrity credibility and reproducibility are preserved. What Are The Major Common Types Of Adaptive Designs Used In Clinical Trials Today.
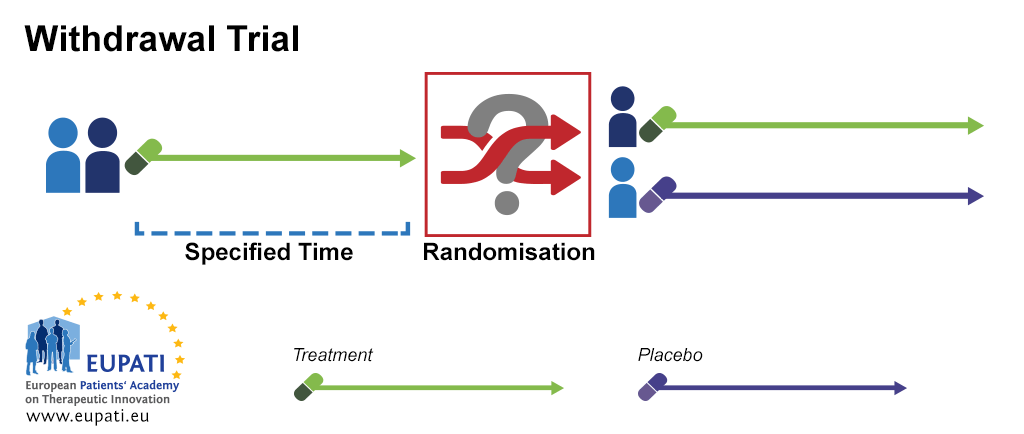 Source: toolbox.eupati.eu
Source: toolbox.eupati.eu
The 33-page guidance which finalizes a draft version released for comment in September 2018 and replaces an earlier guidance from 2010 sets out FDAs recommendations on adaptive trial design principles and the information FDA will review from adaptive studies submitted as part of investigational new drug applications INDs new drug applications. Consequences and gains of possible trial adaptations need to be understood before initiation. The pace of the uptake of adaptive designs in clinical research however has remained well behind that of the statistical literature introducing new methods and highlighting their potential advantages. It provides clarity on how to plan and implement adaptive designs for clinical studies used in medical device development. Clinical Trial Designs Eupati Toolbox.
 Source: pinterest.com
Source: pinterest.com
An adaptive design for a medical device clinical study is defined as a clinical study design that allows for prospectively planned modifications based on accumulating study data without. Stop early for effectiveness futility or safety Bayesian interim monitoring has become an accepted practice for. Analytical results have been derived. Adaptive design for drug and device studies. Pin Na Doske Interface Design.
 Source: pinterest.com
Source: pinterest.com
Consequences and gains of possible trial adaptations need to be understood before initiation. Guidance for Industry and Food and Drug Administration Staff. Adaptive design for drug and device studies. Stop early for effectiveness futility or safety Bayesian interim monitoring has become an accepted practice for. Pin On Farmacevtski Proizvodi.
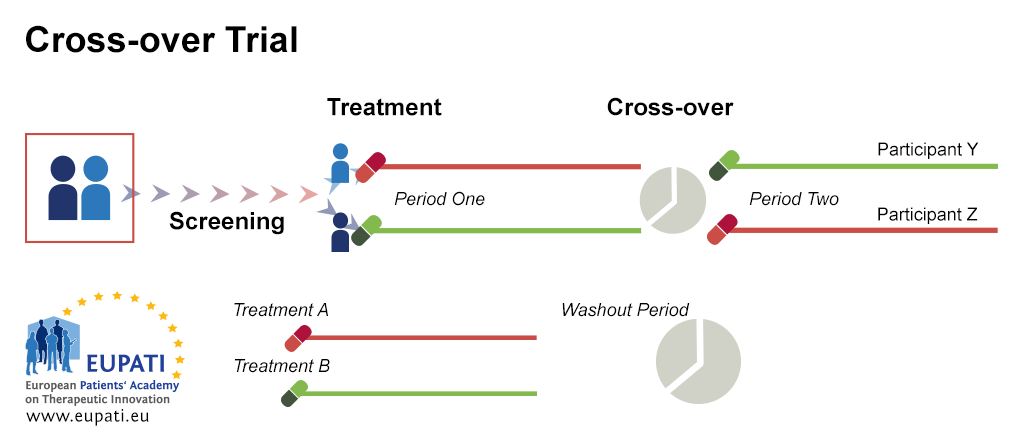 Source: toolbox.eupati.eu
Source: toolbox.eupati.eu
The EMA Reflection paper on methodological issues in confirmatory clinical trials planned with an adaptive design CHMPEWP245902 defines a study design as adaptive if the statistical methodology allows the modification of a design element for example sample-size randomization ratio number of treatment arms at an interim analysis with full control of the. Consequences and gains of possible trial adaptations need to be understood before initiation. An adaptive design is defined as a design that allows modifications to the trial andor statistical procedures of the trial after its initiation without undermining its validity and integrity. What makes a randomised clinical trial adaptive. Clinical Trial Designs Eupati Toolbox.
 Source: pinterest.com
Source: pinterest.com
An adaptive design for a medical device clinical study is defined as a clinical study design that allows for prospectively planned modifications based on accumulating study data without. This final guidance applies to premarket medical device submissions including premarket approval applications PMA premarket notification. Pulm Pharmacol Ther 2010 23. The US Food and Drug Administration FDA on Tuesday finalized guidance that lays out how to design medical device clinical trials that allow for changes based on data while maintaining study validity and integrity. Blink Home Medical Device In 2021 Medical Device Design Devices Design Design Awards.
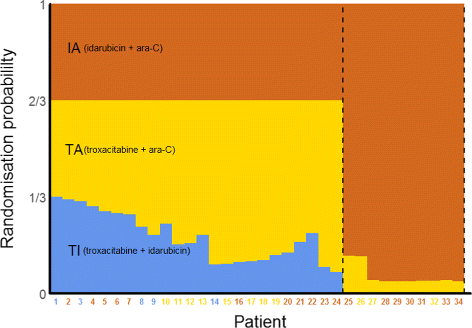 Source: bmcmedicine.biomedcentral.com
Source: bmcmedicine.biomedcentral.com
An adaptive design for a medical device clinical study is defined as a clinical trial design that allows for prospectively planned modifications based on accumulating study data without undermining the trials integrity and validity. It provides clarity on how to plan and implement adaptive designs for clinical studies used in medical device development. Adaptive Clinical Trial Design Case Studies. In particular we resize the study and adjust the critical value for hypothesis testing simultaneously after an interim analysis to achieve a target power and control the type I error rate in comparing AUCs of two modalities. Adaptive Designs In Clinical Trials Why Use Them And How To Run And Report Them Bmc Medicine Full Text.
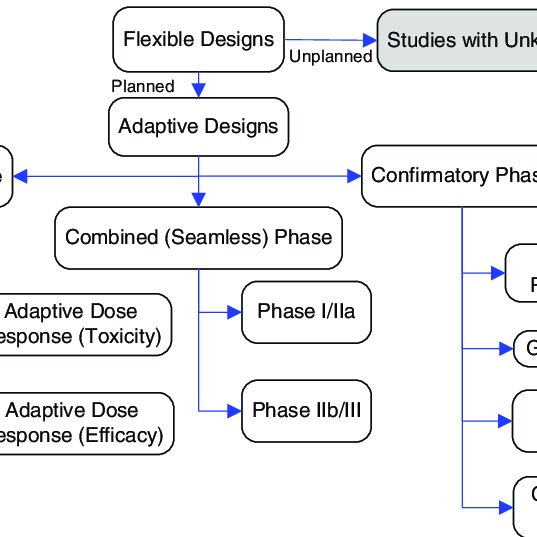 Source: researchgate.net
Source: researchgate.net
Evaluation of medical imaging devices often involves clinical studies where multiple readers MR read images of multiple cases MC for a clinical task which are often called MRMC studies. The US Food and Drug Administration FDA on Tuesday finalized guidance that lays out how to design medical device clinical trials that allow for changes based on data while maintaining study validity and integrity. SMi Groups Adaptive Designs in Clinical Trials Conference will return to London on 3rd and 4th April 2017 and will feature two case studies on Bayesian adaptive designs in medical device development. Today the FDA published the Adaptive Designs for Medical Device Clinical Studies Final Guidance. Summary Of Different Types Of Adaptive Designs For Clinical Trials Download Scientific Diagram.
 Source: jliedu.com
Source: jliedu.com
Bringing in voices from the industrys best clinical specialists strategists and statisticians SMis 9th Adaptive Designs in Clinical Trials conference will feature case. We develop adaptive MRMC design methodologies to enable study resizing. An adaptive design for a medical device clinical study is defined as a clinical trial design that allows for prospectively planned modifications based on accumulating study data without undermining the trials integrity and validity. Adaptive design for medical devices. Adaptive Design Clinical Trials Jli Blog.
 Source: pinterest.com
Source: pinterest.com
Pulm Pharmacol Ther 2010 23. In essence adaptive designs allow prospectively planned modifications to a clinical trial based on interim data provided scientific validity the ability to draw sound inferences and data integrity credibility and reproducibility are preserved. SMi Groups Adaptive Designs in Clinical Trials Conference will return to London on 3rd and 4th April 2017 and will feature two case studies on Bayesian adaptive designs in medical device development. The most frequently appearing type of adaptation was the seamless Phase IIIII design 81142 57 followed by adaptive group sequential 30142 21 biomarker adaptive 28142 20 adaptive dose-finding 23142 16 pick-the-winnerdrop-the-loser 13142 9 sample size re-estimation 11142 8 adaptive randomisation 10142 7 adaptive. 10 Must Haves For Home Health Occupational Therapy Myotspot Com Orthopedics Occupational Therapy Occupational Therapist.
 Source: veristat.com
Source: veristat.com
What is Adaptive Design Clinical Trial. The pharmaceutical landscape is evolving as stakeholders pursue the use of real-world evidence precision medicine and complex innovative trial designs CID to increase efficiency of development. Statistical analysis plans are needed for both interim and final analyses. In particular we resize the study and adjust the critical value for hypothesis testing simultaneously after an interim analysis to achieve a target power and control the type I error rate in comparing AUCs of two modalities. What To Know Before Considering An Adaptive Design Clinical Trial.







